Mercury in the Garden
go.ncsu.edu/readext?824916
en Español / em Português
El inglés es el idioma de control de esta página. En la medida en que haya algún conflicto entre la traducción al inglés y la traducción, el inglés prevalece.
Al hacer clic en el enlace de traducción se activa un servicio de traducción gratuito para convertir la página al español. Al igual que con cualquier traducción por Internet, la conversión no es sensible al contexto y puede que no traduzca el texto en su significado original. NC State Extension no garantiza la exactitud del texto traducido. Por favor, tenga en cuenta que algunas aplicaciones y/o servicios pueden no funcionar como se espera cuando se traducen.
Português
Inglês é o idioma de controle desta página. Na medida que haja algum conflito entre o texto original em Inglês e a tradução, o Inglês prevalece.
Ao clicar no link de tradução, um serviço gratuito de tradução será ativado para converter a página para o Português. Como em qualquer tradução pela internet, a conversão não é sensivel ao contexto e pode não ocorrer a tradução para o significado orginal. O serviço de Extensão da Carolina do Norte (NC State Extension) não garante a exatidão do texto traduzido. Por favor, observe que algumas funções ou serviços podem não funcionar como esperado após a tradução.
English
English is the controlling language of this page. To the extent there is any conflict between the English text and the translation, English controls.
Clicking on the translation link activates a free translation service to convert the page to Spanish. As with any Internet translation, the conversion is not context-sensitive and may not translate the text to its original meaning. NC State Extension does not guarantee the accuracy of the translated text. Please note that some applications and/or services may not function as expected when translated.
Collapse ▲Sources of mercury exposure
Sources outside the garden are typically greater health
risks. Mercury is usually found at very low levels in
soils, but hotspots from man-made sources may also
exist. Below are some of the main sources of mercury
exposure, both in and outside the garden.
Exposure to mercury in the garden
How am I exposed? You can be exposed by eating or breathing contaminated soil particles, or by eating contaminated garden produce.
Are my garden plants safe to eat? Soils tend to contain inorganic mercury. Plants (and people) are not good at absorbing mercury in this form. Garden produce is not a major source of mercury exposure.
Should I be worried? It is important to remember that there are many health benefits associated with gardening. Mercury exposure is a serious concern in mining communities and among communities that eat certain types of fish. However, garden exposure at high levels is uncommon. Even so, limiting exposure, especially for children, is always a good idea.
Limit children’s exposure
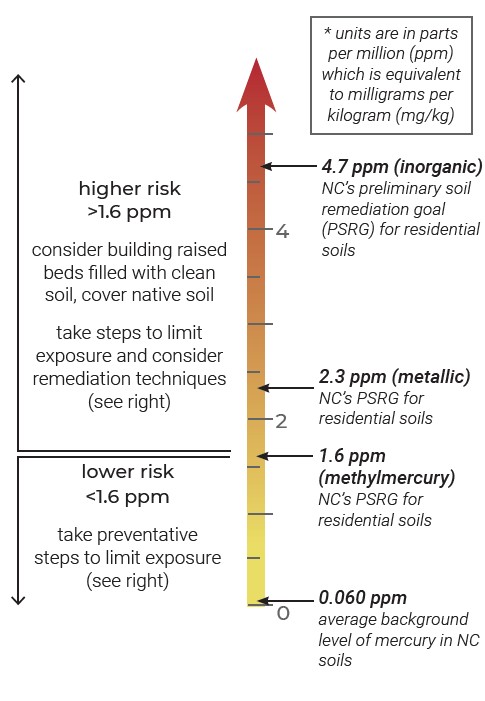
Making sense of regulatory standards
No official standards have been established in North Carolina for acceptable levels of mercury in garden soils. For remediating residential soil, North Carolina uses EPA guidelines for different forms of mercury. New York State’s guideline (0.81 ppm*), considers exposures like gardening and assumes mercury is in its most toxic form, so it is a conservative value that represents an abundance of caution.
Health impacts of mercury
Exposure to inorganic mercury at high levels can damage kidneys, stomach, and intestines. Impacts to the nervous system can cause mood swings, memory loss, and behavioral changes. Skin exposure can cause rashes and dermatitis.
Children and pregnant women are at greatest risk from mercury exposure. The developing fetus is vulnerable to the toxic effects of maternal mercury exposure.
Methylmercury is particularly toxic and can accumulate up the food chain, but it is not usually found in garden soils.
Reduce mercury exposure in the garden
- Uptake of mercury by plants is low, so focus on controlling dust and limiting soil ingestion
- Remove boots or shoes after gardening to reduce the amount of contaminated soil you track into your home
- To reduce mercury particles in air from dust, cover bare soil with mulch and keep the soil moist
- Flooded soils may help convert mercury to its more toxic form, but this can take many months
- Adding compost or other organic matter from a contaminant-free source may help dilute mercury concentration in soil. Check the NC Composting Council website to find STA or OMRI certified compost
- Conduct a soil safety training for all garden users on exposure reduction strategies
- Visit our website below for our factsheet on 10 Healthy Garden Habits
Testing Resources
 |
How to test your soil and interpret the results |
 |
More Information |
 |
Still have questions about mercury soil testing? Email us at superfund@duke.edu. |
Email us at superfund@duke.edu
This factsheet was created by the Duke University Superfund Research Center’s Community Engagement Core with the goal of helping garden managers, Extension agents, Master Gardeners, and home gardeners identify, understand, and manage risks associated with chemical contamination that may be present in garden soils. This work was supported through the National Institute of Environmental Health Sciences P42 Multiproject Center Grant program, grant number P42ES010356.









